Migraine is not just a pain the head. It is accompanied by other sensory—visual, auditory and olfactory—hypersensitivities and cutaneous allodynia. The complex network of neural structures and pathways involved in pain sensation and processing and the migraine pain experience were described by Professor Todd J. Schwedt, Mayo Clinic, Phoenix, AZ, at the Virtual Scottsdale Headache Symposium 2020.
The trigeminocervical system
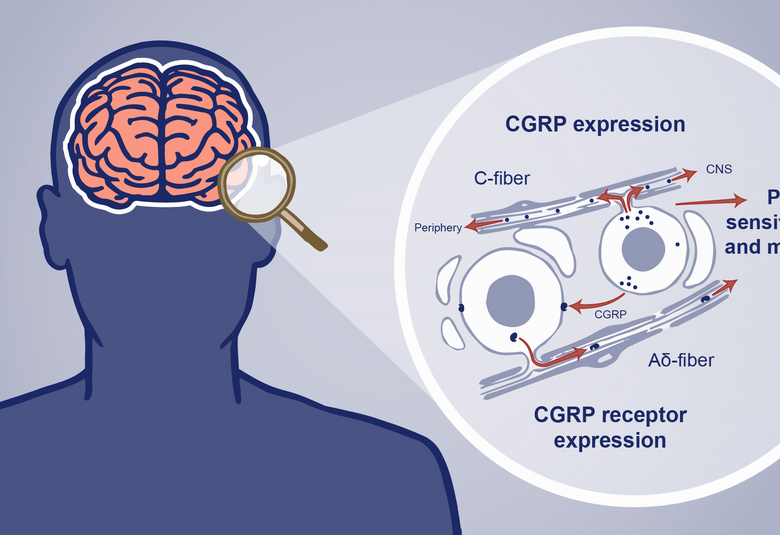
Activation of trigeminal and occipital nerve sensory afferents leads to the release of vasoactive CGRP
The trigeminocervical system is the anatomic system responsible for head pain including migraine, said Professor Schwedt. It includes:
- the trigeminal nerve
- the trigeminal ganglion (TG)
- the trigeminal nucleus caudalis (TNC) of the brain stem
- C1 and C2 segments of the cervical spine1,2
Facial sensation is provided by the trigeminal nerve through its ophthalmic (V1), maxillary (V2), and mandibular (V3) branches. V1 also provides sensation for the proximal portions of major intracranial arteries and meningeal arteries.1,2 The trigeminal nerve projects to the TG and thence to the TNC,1,2 explained Professor Schwedt.
The sensation of pain is made up of multiple different components
Sensation from the back of the head is provided by the second cervical nerve through the greater and lesser occipital nerves.1,2
Activation of trigeminal and occipital nerve sensory afferents leads to the release of vasoactive neuropeptides including calcitonin gene-related peptide (CGRP) into the blood. These neuropeptides:
- lead to neurogenic inflammation with arterial vasodilation
- act as neurotransmitters to facilitate the pain experience1,2
Does migraine result from enhanced pain facilitation and reduced pain inhibition?
Ascending and descending pathways in the central nervous system (CNS) facilitate and inhibit pain, respectively,2 and it has been suggested that migraine might result from enhanced pain facilitation and reduced pain inhibition, said Professor Schwedt.
Pain processing and the pain experience
Ascending fibers from the TNC carry sensory input to the hypothalamus and thalamus.2 Further ascending fibers from the thalamus project to multiple regions of the cortex—the so-called pain matrix—which are active during the pain experience regardless of the type of pain.2,3
Pain characteristics depend upon which areas of the brain are involved in the pain experience
The sensation of pain is therefore made up of multiple different components, and the characteristics of the pain depend upon which areas of the brain are involved, said Professor Schwedt.
- “Sensory discriminative” pain describes pain type, intensity, and location, and involves the primary sensory cortex and the posterior insula.
- “Affective-motivational” pain describes emotional aspects such as fear and involves the anterior insula, anterior cingulate cortex, and amygdala.
- “Cognitive” pain describes cognitive aspects such as attention or distraction and involves the prefrontal cortex and hippocampus.
- “Integrative” pain describes the integration of pain modalities and multisensory integration and involves the temporal pole and the temporal parietal junction.3
Atypical multisensory integration has been observed in migraine
Multisensory integration appears to play an important role in migraine with symptoms arising from multiple sensory domains—eg, headache, cutaneous allodynia, visual and auditory sensitivity,3 added Professor Schwedt. Furthermore, in migraine:
- atypical multisensory integration has been observed, eg, exposure to bright light worsens the headache
- interictal photosensitivity has been linked to altered brain excitability and altered brain structure4,5
Our correspondent’s highlights from the symposium are meant as a fair representation of the scientific content presented. The views and opinions expressed on this page do not necessarily reflect those of Lundbeck.